|
The term bonding groups/domains (second from the left column) is used in the columnįor the bonding pair of electrons. On an individual atom that are not shared with another atom. Non-bonding pairs of electrons are those pairs of electrons Of electrons are those electrons shared by the central atom and any atom to Of bonding pair of electrons and non-bonding pairs of electrons. 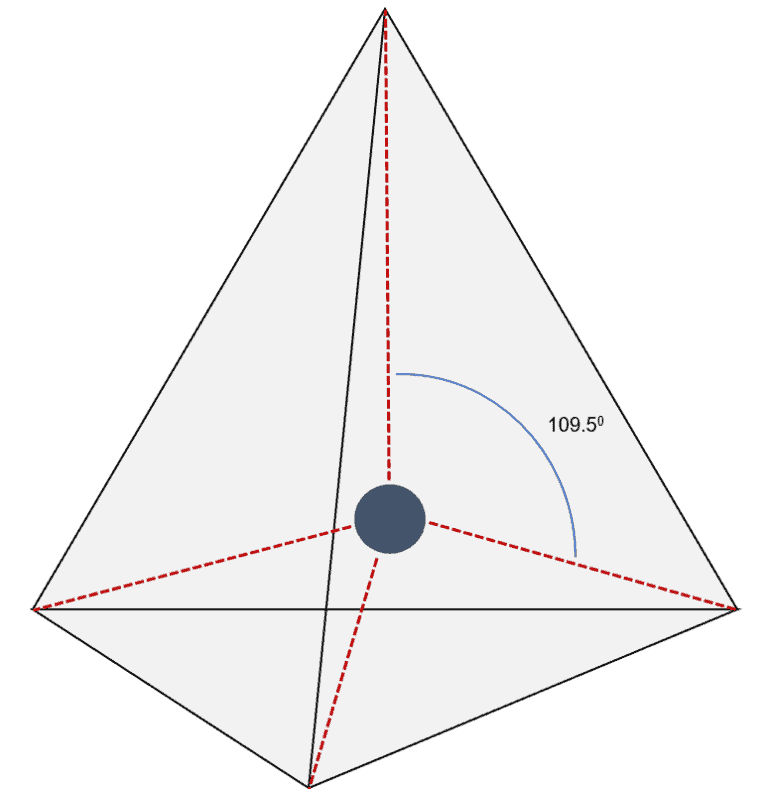
The table below contains several columns. To use the model we will have to memorize a collection Model called the Valence Shell Electron-Pair Repulsion (VSEPR) model that isīased on the repulsive behavior of electron-pairs. Of simple molecular (covalent) compounds and polyatomic ions. The usual bond angle of 109.6 degrees is not the same and it is reduced to 104.5 degrees.Molecular Geometry Molecular Geometry VSEPR At this point we are ready to explore the three dimensional structure This variant is the deformed state of tetrahedral geometry and thus it is called the bent geometry. One lone pair is in the plane coming out from the screen, the other lone pair is placed on the screen entering into the screen, and thus two single bonds are in the same plane of the screen. On the other side, the bond angles between two single bonds will decrease. The lone pair will repel single or double covalent bonds formed adjacent to itself and thus it increases the bond angle from its side. We have read that a lone pair will affect the overall geometry along with some alterations in the bond angle. Since the lone pairs are coming into action in this variant type. This variant comes along with AX2E2 (A=central atom, X= electron acceptor, and E = lone pair) when there are two different types of acceptor atoms and they have different covalency. When there is some lone pair: – This three-dimensional molecule is formed when the four valence electrons of the central atom get distributed to two other acceptor atoms, and the remaining electrons are contributing to the formation of lone pair.There are four vertices and six edges in a tetrahedron.Ī tetrahedron is actually resembling solid by the help of which we can easily understand what is a tetrahedral geometry and its properties. A tetrahedron is also called a pyramid with a triangular base. Each face of a tetrahedron is an isosceles triangle. What Is Tetrahedral Geometry?Ī tetrahedron is a three-dimensional closed shape made up of four faces. In this article, you’ll learn what is the tetrahedral shape of a molecule and what are some compounds that exist in the tetrahedral shape. The tetrahedral geometry is widely seen in the molecules, and it also has different bond angles. The molecular structure of a three-dimensional molecule doesn’t change rapidly and is found in nature in the same orientations. Molecular geometry is the name of the study that is done on different atoms that combine with one primary atom at the center by bonding and forming a specific physical structure. When we combine the meaning of both these terms, we know that tetrahedral means a solid with four faces. 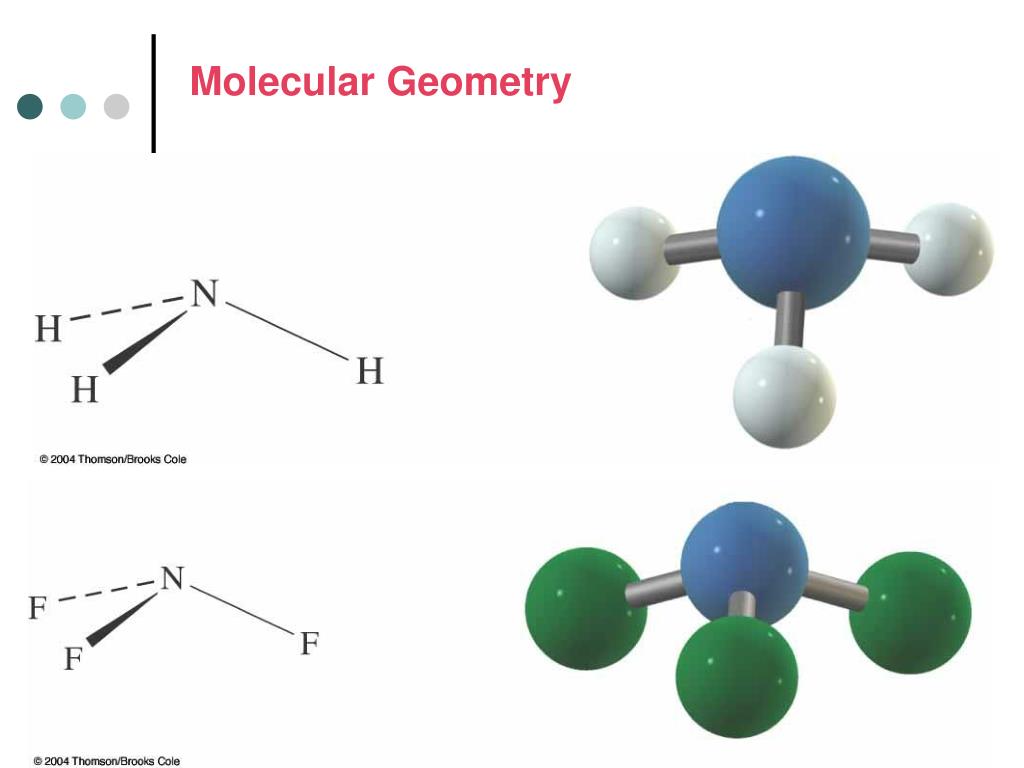
“Tetra” means the number four, and the word “hedral” depicts a solid face. By the word “tetrahedral,” we can get a slight idea about the meaning of this term. The tetrahedral shape is formed when four atoms in their elemental form join together by covalent bonding.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
